

Floor map of the Mineralogy Museum

The entrance room displays some of the most beautiful specimens in the museum. Theses specimens come from all around the world and include the world-renowned franklinite (6279), the optical calcite (6269), a gigantic molybdenite (6236), a colorful pink spodumene (kunzite, 8560), gypsums with incredible shapes, pink and green beryls, a spectacular Moroccan erythrite (6287), an aesthetic sepiolite (6280), and even the first artificial ruby (6302) created in 1870 by Verneuil.
(Display case A3, sample #6298, from Eisenerz, Tyrol, Austria)
This strangely shaped mineral, which was originally considered a coral, was described in 1748 by John Hill and in 1768 was named “Flos Ferri”, “iron flower” in latin, by Linné. In 1801, Haüy described it as a coralloid carbonated lime.
The typical deposit from which this type of specimen comes is from one similar to the iron mine of Eisenerz in Tyrol, Austria (Eisenerz means iron mine in German). In Eisenerz, the “flos ferri” is found in cavities of carbonated iron ore (siderite).
These twisted and branched crystallizations are also present in some caves; in this case they are called helictites.

(Display case A8, sample #6269, from Eskifjord, Island)
In the middle of the 17th century, a large amount of optical calcite (CaCO3) was shipped to Denmark from Iceland. Mineralogists quickly appreciated its curious property of doubling images. This phenomenon of double refraction was described by Erasme Bartholin in 1669, and later explained by Huyghens in his « Treaty on Light ». Opticians later used optical calcite to create polarizers of better quality than those used prviously.
From the 17th century onwards, the deposit of Eskefiord was rapidly exploited. The deposit was made of a large cavity measuring 15 x 4 x 3 meters, mostly filled with large transparent crystals of calcite. The largest crystal in the cavity was 6-meter long by 2-meter large. It is possible that the calcite cleavage in the museum is the top part of this crystal. The tarnished faces of the specimen are the natural faces of the original crystal. On some of them, one can observe small crystals of stilbite, which is a common zeolite in basaltic cavities. The shiny faces represent the cleavage planes that appeared during the extraction due to mechanical breakage of the original large crystal. In this specimen, the phenomenon of double refraction is easily seen through the parallel cleavage faces.
Since 1930, polarizers have been built in a much cheaper way using Polaroid slices. Polaroid is a material made of silicate or organic glass that contains microcrystals of heraphite (iodoquinine sulfate), which was invented by Edwin H. Land. Interestingly, the microcrystals are highly dichroic and are all oriented in the same direction.

(Display case A12, sample #6278 from Lake Superior, Ontario, Canada)
Native copper from the south bank of Lake Superior was mined by Chipeway Indians, using stone axes, long before the arrival of Europeans on the American continent. It was used to make jewelry, arrow heads, and tobacco pipes. A giant copper nugget rediscovered on the Ontonagon River in 1819 was used as primary source for centuries. Its remains are now preserved at the Smithsonian Institution in Washington, DC.
Geologic explorations conducted between 1820 and 1840 found crystallizations of native copper trapped inside calcite geodes contained in basaltic flows that cover the area. An important mining company was created in 1846 in order to exploit this priceless natural resource.

(Display case A12, sample #6279)
Franklinite was first found in Franklin Furnace, New Jersey, USA, at the end of the 18th century. It was originally thought to be bad iron ore. It was named by the mineralogist Berthier in 1819 after the Franlkin Furnace mine. This mine as well as the Sterling Hill mine, its neighbor, contain an extraordinary diversity of mineral species, with over 300 species recognized, including the main species franklinite, willemite and zincite, which made the deposit famous.
The large octahedron of franklinite found in this specimen is known to be the largest in the world. Broken during its extraction, it was repaired by Claude Guillemin, a former curator of the Museum. In the Haüy room (Room B), one can observe the spectacular reaction when franklinite is exposed to ultraviolet light. The mineral luminesces bright green, while its associated calcite luminesces bright orange.

(Display case A9, sample #13644, from Terlingua, Texas, USA)
Often described as somewhat misshapen stalagmites or stalactites, these curious "flowers" of gypsum have a very different structure. Unlike stalagmites or stalactites they have no channel in their center and have almost a single crystal structure. This suggests that the mechanism of formation is quite different.
Originally, gypsum deposits are formed in lagoons or shallow seas as sea water evaporates. Depending on the geological setting, gypsum may crystallize in many forms. In old abandoned mines, the circulation of water amongst the rocks allows water borne bacteria to attack and break down sulfide ores. Sulfate-rich water can then percolate and evaporate on the walls of galleries, leaving behind this kind of ram’s horn gypsum. Growing by the base, just like a hair, gypsum crystals appear in only a few tens of years.
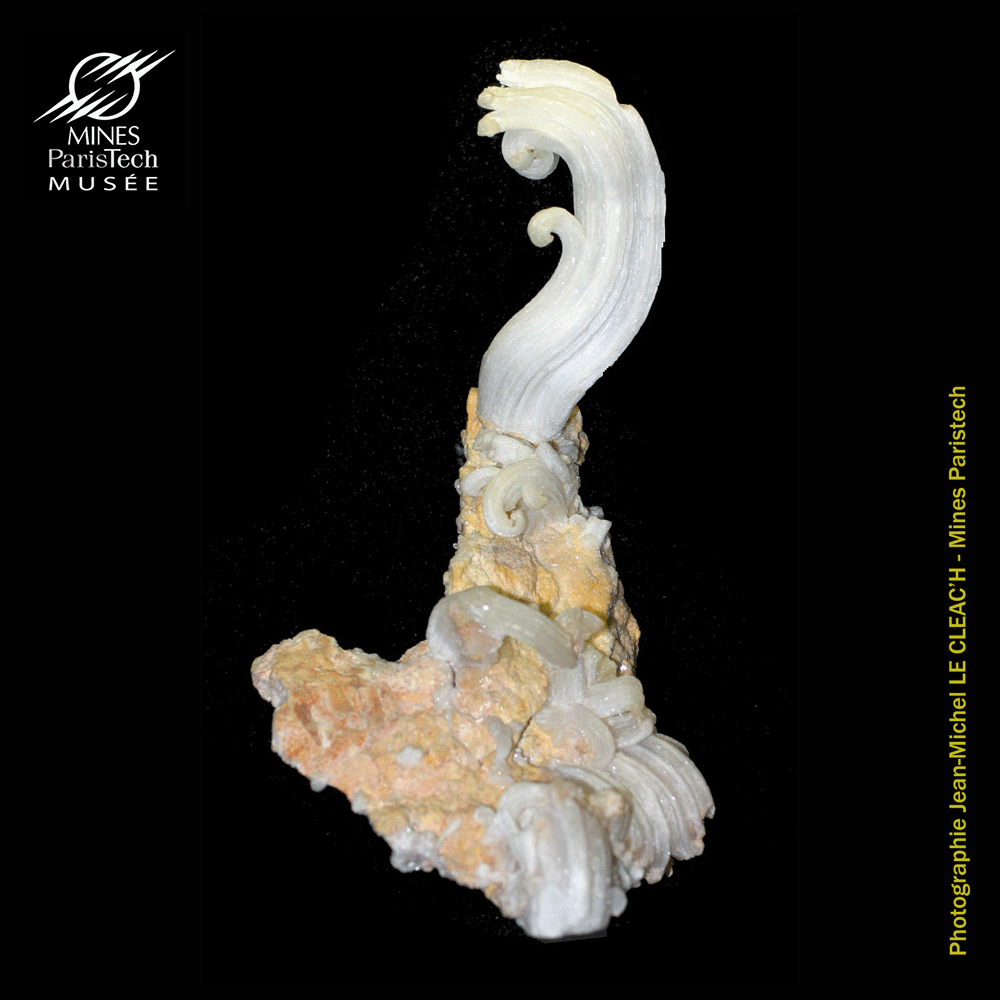
(Display case A6, sample #16266, from Kakanda, Domican Republic of Congo)
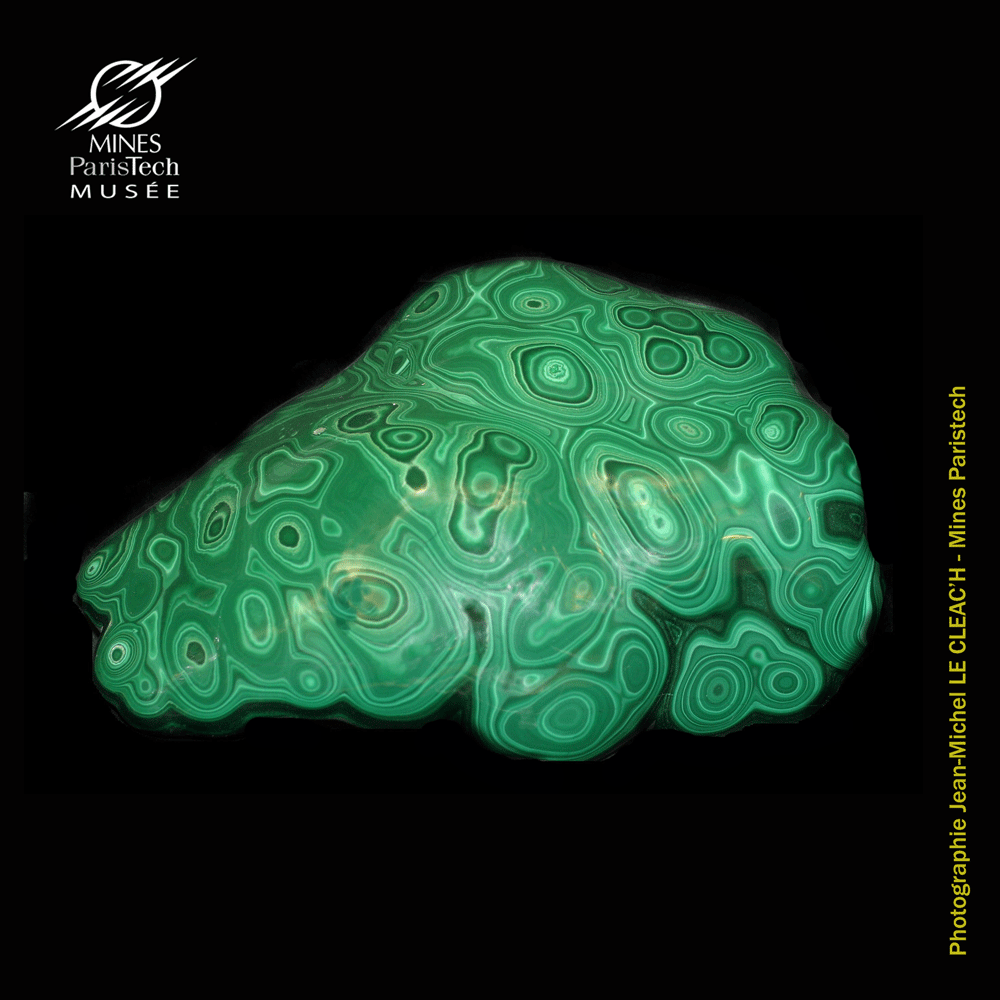
(Display case A13, sample #15580, from Hannan's Brown Hill, South Australia, Australia)

(Display case A11, sample #16535, from Brejinho das Ametistas, Bahia, Brazil)

(Display case A10, sample #6273, from the Alps)

(Display case A3, sample 6302)

(Display case A1, sample #6280 from Sari Sou, Yarimca, Turkey)
Any questions about the museum or the collection ? Contact us
Find all the information to come to the Mineralogy Museum here
Musée de Minéralogie
Mines Paris - PSL
60 boulevard Saint Michel
75006 Paris
OPENING HOURS FROM AUGUST 13th:
TUESDAY: 10am - 12pm and 1:30pm - 6pm.
WEDNESDAY, THURSDAY, FRIDAY: 1:30pm - 6pm
SATURDAY: 10am - 12:30pm and 2pm - 5pm
Closed on National Holidays, Sundays and Mondays
NEXT CLOSING DAYS:
Exceptionnally closed on Saturday August 17, 2024
Mines Paris - PSL will be closed during the Olympic Games. For this reason, the museum will also be closed from SATURDAY JULY 20 UNTIL MONDAY AUGUST 12, 2024.
Find all our temporary exhibits and special events here
